|
The most important absorption reactions are divided by the exit channel into two following reactions: Therefore a variety of emissions or decays may follow. This is a very important feature because the decay mode of such a compound nucleus does not depend on how the compound nucleus was formed. The absorption reactions are reactions where the neutron is completely absorbed, and the compound nucleus is formed. The neutron absorption reaction is the most important type of reactions that take place in a nuclear reactor. Neutrons may interact with nuclei in one of the following ways: It can be calculated from the following equation: In short, neutrons collide with nuclei, not with atoms. A very descriptive feature of the transmission of neutrons through bulk matter is the mean free path length ( λ – lambda), which is the mean distance a neutron travels between interactions. 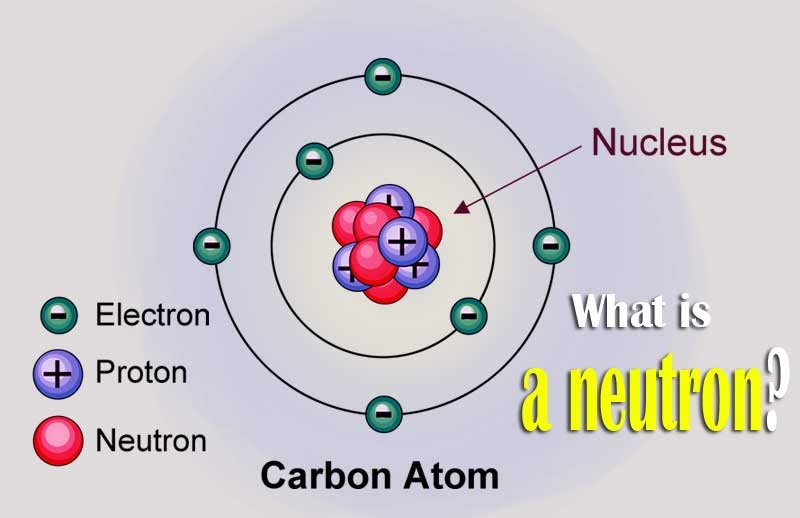
Neither the electrons surrounding (atomic electron cloud) a nucleus nor the electric field caused by a positively charged nucleus affect a neutron’s flight. Therefore they travel in straight lines, deviating from their path only when they collide with a nucleus to be scattered into a new direction or absorbed. Most absorption reactions result in the loss of a neutron coupled with the production of one or more gamma rays since the resulting nucleus is usually unstable. For example, when neutrons strike the hydrogen nuclei, proton radiation (fast protons) results. production in the atmosphere by cosmic ray collisions).Neutrons have zero electrical charges and cannot directly cause ionization. 
Thus any tritium we observe in nature must have been produced very recently in energetic events (e.g. That too can be formed in minute quantities in the big bang and inside stars or by spallation reactions, but here there is the additional problem that any tritium produced is unstable with a half-life of 12.3 years. Similar arguments pertain to the tritium (one proton, two neutrons) form of hydrogen. Here the issue is that disintegration to produce alpha particles and free neutrons is more energetically favoured. For similar reasons it is very difficult to produce deuterium by fission (e.g. That would require some non-equilibrium process that produced deuterium-enriched material and then cooled it on short timescales. Thus there is very little deuterium in the universe because its production by fusion requires a temperature high enough to then allow further fusion to produce the more energetically favourable helium. The net effect of stellar nucleosynthesis is therefore to destroy deuterons - both those produced in stellar nucleosynthesis and those produced in the early universe. As such, a significant fraction (maybe 10-20%) of the hydrogen in most stars does get transformed (briefly) into deuterium during their main-sequence lifetimes, but again the deuterons are then quite unstable to subsequent reactions that rapidly synthesise helium. The formation of deuterons is the first stage in the pp-chain of nuclear reactions that turn hydrogen into helium. Protons can get subsequently converted into deuterons during nuclear fusion in stellar cores. Thus the vast majority of hydrogen is in the form of its neutronless isotope.  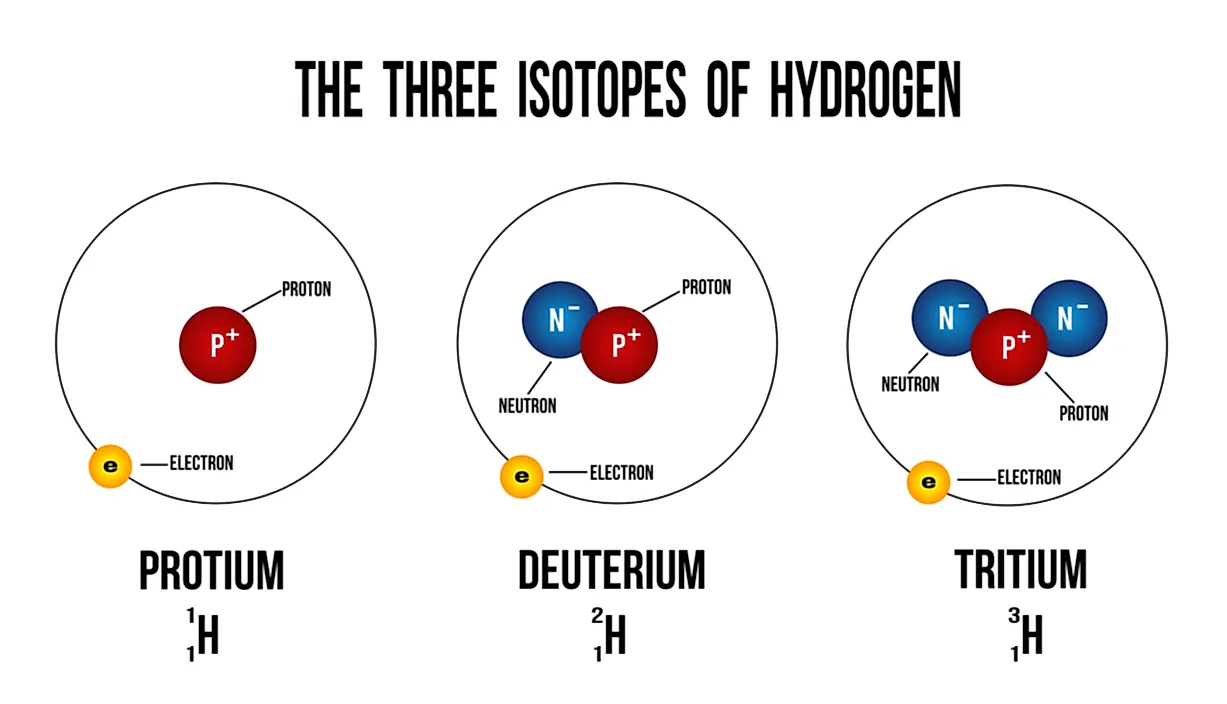
However, the system is in thermal equilibrium, thus seeking to minimise the total energy density although the deuterons are stable, helium is a much more stable nucleus and as a result the deuterons rapidly combine to form helium and this mops up all the available neutrons in the early universe. The answer is that they do - there is a brief window of time where deuterons can form and the universe is cool enough that they don't immediately break apart. Your question boils down to why didn't protons in the very early universe combine with neutrons to form deuterons (a proton plus a neutron).
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
